  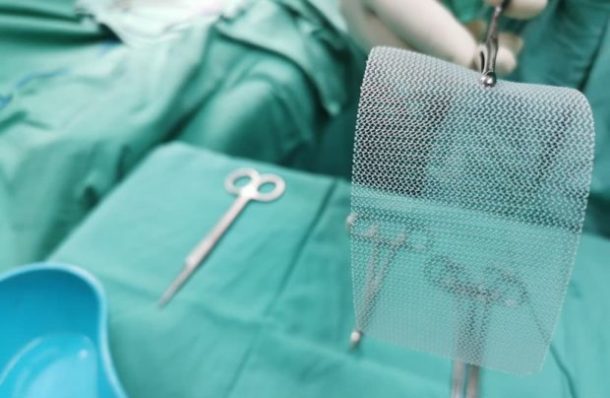
The known complications associated with defective hernia mesh products include:Īdditionally, patients harmed by a defective hernia mesh often must undergo repeated surgeries to remove the device. The suit claims the C-QUR Mesh’s Omega 3 gel coating made from fish oil is a major defect in the design of the implant, because it can cause the implant to curl and deform over time, leading to serious inflammation and injuries. As a result of the mesh and the consequent removal of the implant, the plaintiff allegedly missed six weeks of work and develop scar tissue buildup in her abdomen. District Court for the Middle District of Georgia. One Georgia woman suffered seroma (a buildup of fluids in the body) and nerve damage to the abdomen after receiving a defective C-QUR mesh implant, according to the lawsuit Bryant v. The suit claims that Physiomesh as a product is defective, prone to becoming embedded in human tissue over time, and is “inappropriately designed and engineered for use in hernia repair,” creating an unreasonable risk of injury to patients.Ītrium C-QUR Mesh Lawsuits Allege the Mesh’s Gel Coating Is Defective District Court for the Middle District of Florida. While the actual settlement amount has yet to be released, it was reported that the company set aside 66 million. In a huge win for over 1,300 claimants, Atrium Medical Corp, a Swedish medical device manufacturer, has agreed to settle hernia mesh lawsuits filed against it in the New Hampshire courts. 
There are currently about 20 Physiomesh and 24 C-QUR hernia mesh lawsuits filed in federal court.Įthicon’s Physiomesh Lawsuits Allege the Product Caused Permanent Injuriesĭoctors implanted a Florida woman with an Ethicon Physiomesh flexible composite mesh during hernia repair surgery and she suffered permanent injuries as a result, according to the lawsuit Quinn v Ethicon, Inc. Atrium Settles C-QUR Hernia Mesh Lawsuits. Patients harmed by a defective hernia mesh often must undergo repeated surgeries to remove the device.Ī hernia mesh is a medical device used to support weakened or damaged tissue following a surgery to repair a hernia.īoth Atrium’s C-QUR and Ethicon’s Physiomesh hernia mesh implants have reportedly caused serious injuries in patients who underwent hernia surgery - ranging from pain and infection to adhesion to tissue and bowel obstruction. What is a Hernia Mesh and Why Are People Reporting Problems with It? Contact us to learn if you could be eligible for compensation for suffering from these serious injuries. If you have received surgical mesh during a recent hernia repair surgery, you could be at risk of adverse medical effects and complications including bowel obstruction, mesh adhesion to tissue, hernia recurrence, and more. Our attorneys are currently investigating claims that certain hernia surgical mesh implants made by medical manufacturing companies Atrium Medical and Johnson & Johnson subsidiary Ethicon are defective and have caused serious injuries to hernia surgery patients. Hernia Mesh The JPML Met Last Week Heres What Theyve Decided New Bellwethers Scheduled in Bard Hernia Mesh MDL 2846, With Many Expecting a Settlement Soon. Atrium C-QUR, Ethicon Physiomesh Hernia Mesh Allegedly Cause Severe Intestinal Complications 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
